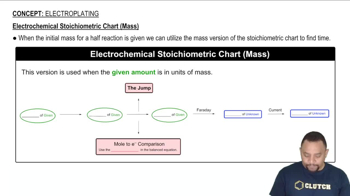
20. Electrochemistry
Electroplating
Problem 96b
Textbook Question
Textbook QuestionA mixture of copper and gold metals that is subjected to electrorefining contains tellurium as an impurity. The standard reduction potential between tellurium and its lowest common oxidation state, Te4+, is Te4+1aq2 + 4 e- ¡ Te1s2 E°red = 0.57 V Given this information, describe the probable fate of tellurium impurities during electrorefining. Do the impurities fall to the bottom of the refining bath, unchanged, as copper is oxidized, or do they go into solution as ions? If they go into solution, do they plate out on the cathode?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
408
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos