8. Thermochemistry
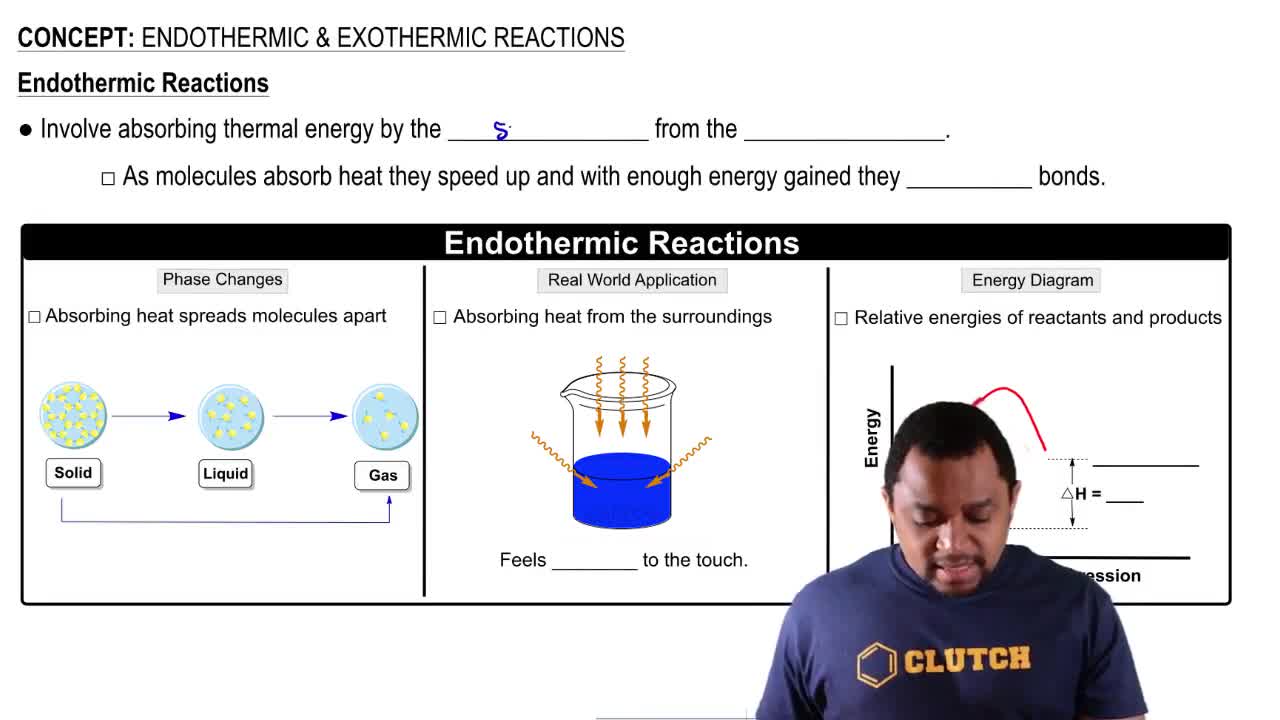
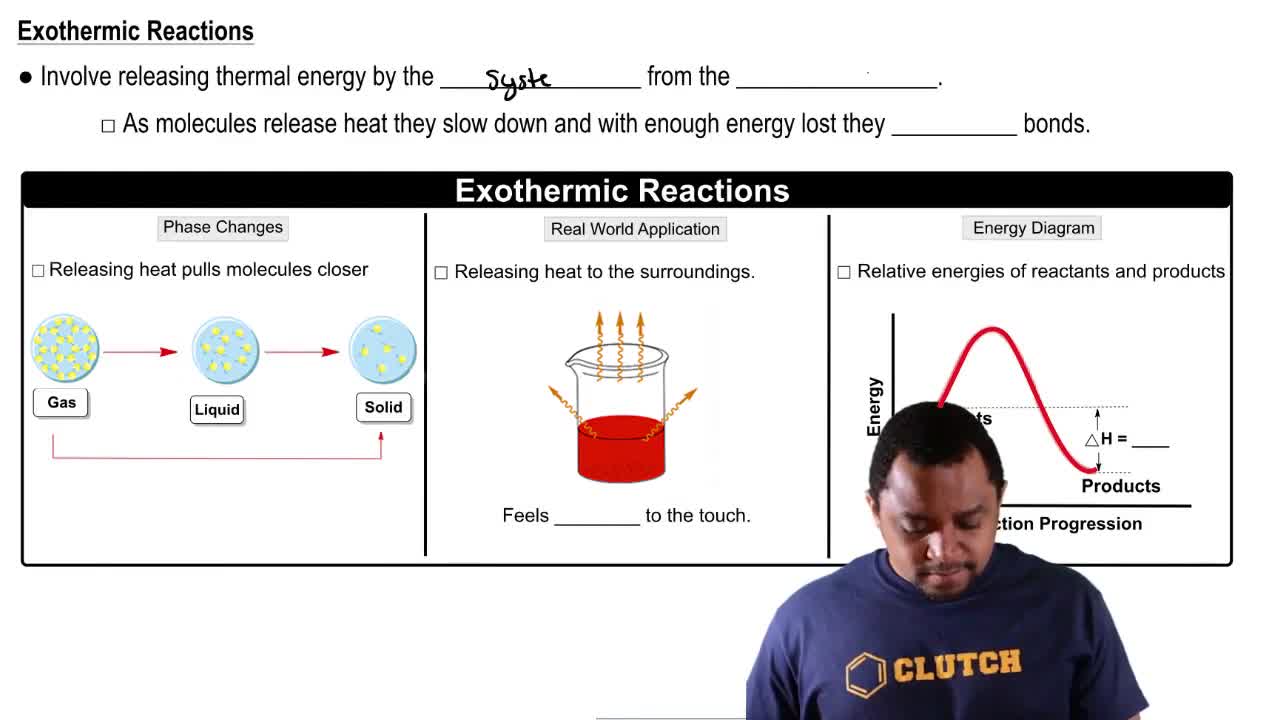
Endothermic & Exothermic Reactions
Problem 96
Textbook Question
Textbook QuestionAn important reaction for the conversion of natural gas to other useful hydrocarbons is the conversion of methane to ethane. 2 CH4(g) → C2H6(g) + H2(g) In practice, this reaction is carried out in the presence of oxygen, which converts the hydrogen produced into water. 2 CH4(g) + 12 O2(g) → C2H6(g) + H2O(g) Use Table 8.3 to estimate H for these two reactions. Why is the conversion of methane to ethane more favorable when oxygen is used? Why is the conversion of methane to ethane more favorable when oxygen is used?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
582
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos