7. Gases
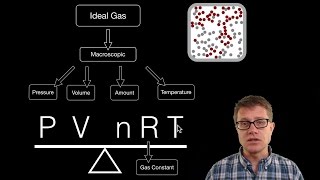
The Ideal Gas Law
Problem 138
Textbook Question
Textbook QuestionWhen solid mercury(I) carbonate, Hg2CO3, is added to nitric acid, HNO3, a reaction occurs to give mercury(II) nitrate, Hg1NO322, water, and two gases A and B: Hg2CO31s2 + HNO31aq2¡ Hg1NO3221aq2 + H2O1l 2 + A1g2 + B1g2 (a) When the gases are placed in a 500.0-mL bulb at 20 °C, the pressure is 258 mm Hg. How many moles of gas are present?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
582
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos