6. Chemical Quantities & Aqueous Reactions
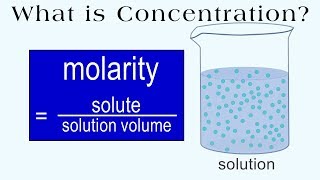
Molarity
Problem 153
Textbook Question
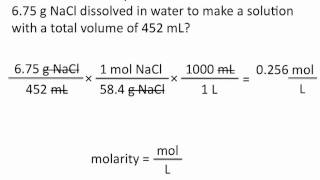
Textbook QuestionA railroad tank car derails and spills 36 tons of concen-trated sulfuric acid. The acid is 98.0 mass% H2SO4 and has a density of 1.836 g/mL. (a) What is the molarity of the acid?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
392
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos