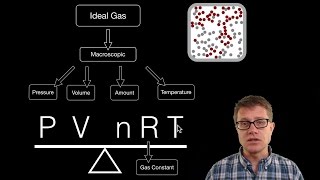
7. Gases
The Ideal Gas Law
Problem 1
Textbook Question
Textbook QuestionAt 273 K and 1 atm pressure, 1 mol of an ideal gas occupies 22.4 L. (Section 10.4) (b) Looking at Figure 18.1, we see that the temperature is lower at 85 km altitude than at 50 km. Does this mean that one mole of an ideal gas would occupy less volume at 85 km than at 50 km? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
3188
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos