6. Chemical Quantities & Aqueous Reactions
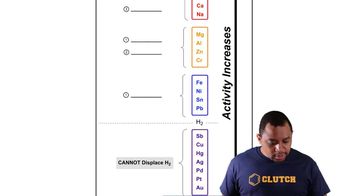
Activity Series
Problem 98c
Textbook Question
Textbook QuestionBronze is a solid solution of Cu(s) and Sn(s); solutions of metals like this that are solids are called alloys. There is a range of compositions over which the solution is considered a bronze. Bronzes are stronger and harder than either copper or tin alone. (c) Suggest a reaction that you could do to remove all the tin from this bronze to leave a pure copper sample. Justify your reasoning.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
928
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos