12. Molecular Shapes & Valence Bond Theory
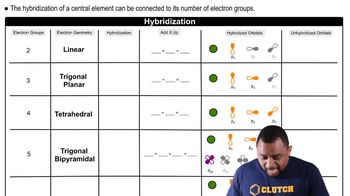
Hybridization
Problem 61c
Textbook Question
Textbook QuestionConsider the Lewis structure for acetic acid, which is known as vinegar: (b) What are the hybridizations of the orbitals on the two oxygen atoms, and what are the approximate bond angles at the oxygen that is connected to carbon and hydrogen? What are the hybridizations of the orbitals on the two oxygen atoms?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
950
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos