11. Bonding & Molecular Structure
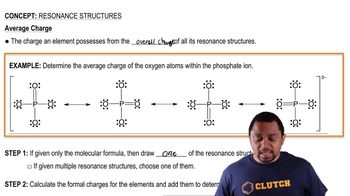
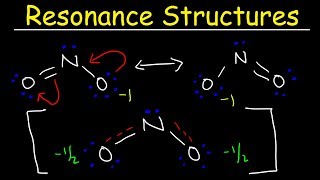
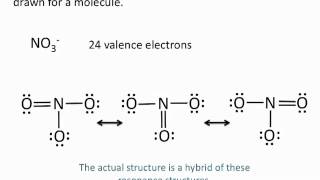
Resonance Structures
Problem 97
Textbook Question
Textbook QuestionThe cyanate ion (OCN- ) and the fulminate ion (CNO- ) share the same three atoms but have vastly different properties. The cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. The resonance structures of the cyanate ion are explored in Example 9.8. Draw Lewis structures for the fulminate ion—including possible resonance forms— and use formal charge to explain why the fulminate ion is less stable (and therefore more reactive) than the cyanate ion.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
2613
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos