13. Liquids, Solids & Intermolecular Forces
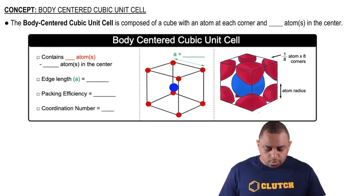
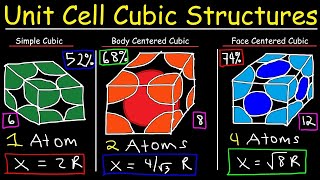
Body Centered Cubic Unit Cell
Problem 83
Textbook Question
Textbook QuestionAn unknown metal is found to have a density of 7.8748 g/cm3 and to crystallize in a body-centered cubic lattice. The edge of the unit cell is 0.28664 nm. Calculate the atomic mass of the metal.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
2528
views
2
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos