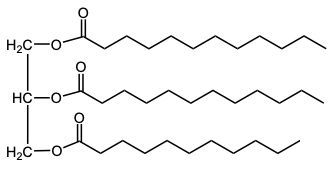
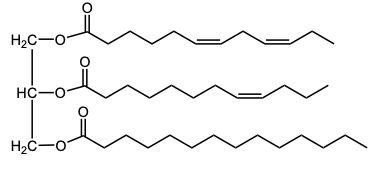
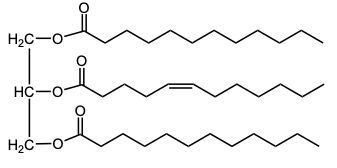
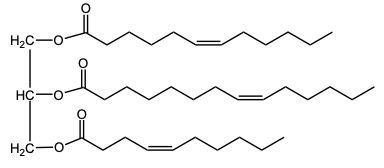
In the oxidation of triacylglycerols, elemental oxygen (O2) plays a crucial role by cleaving the pi bonds present in the triglyceride structure. This reaction transforms the double-bonded carbons into carboxylic acids. Initially, the triglyceride molecule contains multiple pi bonds, which are susceptible to oxidation. When atmospheric oxygen interacts with these pi bonds, it effectively breaks them apart.
As a result of this cleavage, each of the double-bonded carbons is converted into a carboxylic acid. This transformation involves the formation of a double bond between the carbon and an oxygen atom, along with the addition of a hydroxyl group (–OH) to create the carboxylic acid functional group. The general structure of a carboxylic acid can be represented as RCOOH, where R denotes the hydrocarbon chain derived from the original triglyceride.
Thus, the oxidation of triacylglycerols leads to the generation of two carboxylic acids from the cleavage of pi bonds, highlighting the significance of environmental oxygen in this biochemical process. Understanding this reaction is essential for comprehending lipid metabolism and the biochemical pathways involved in fatty acid oxidation.