1. Intro to General Chemistry
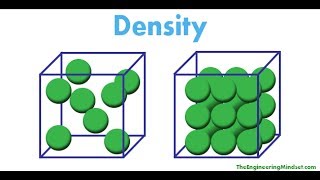
Density
Problem 37a
Textbook Question
Textbook Question(a) To identify a liquid substance, a student determined its density. Using a graduated cylinder, she measured out a 45-mL sample of the substance. She then measured the mass of the sample, finding that it weighed 38.5 g. She knew that the substance had to be either isopropyl alcohol 1density 0.785 g>mL2 or toluene 1density 0.866 g>mL2. What are the calculated density and the probable identity of the substance?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1307
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos