23. The Second Law of Thermodynamics
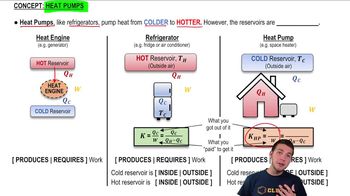
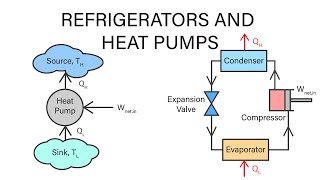
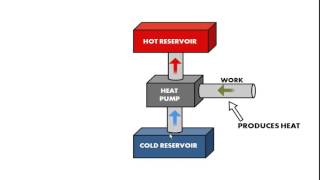
Refrigerators
Learn with other creators
Practice this topic
- Multiple Choice
A refrigerator has a coefficient of performance of 2.4. Each cycle, it takes in 3×104 J of heat from the cold reservoir. How much is expelled to the hot reservoir?
1531views1rank - Multiple ChoiceFreezing a kilogram of water, initially at 20°C, requires your refrigerator to remove about of thermal energy from the water. If the coefficient of performance of your refrigerator is 5.0, how much heat is exhausted into your kitchen when you freeze a kilogram of water?1759views
- Multiple ChoiceWhat is the maximum possible coefficient of performance for a refrigerator operating between 2.0°C (a typical refrigerator temperature) and 20°C (a typical kitchen temperature)?1594views
- Textbook Question
A certain brand of freezer is advertised to use kWh of energy per year. What is the theoretical maximum amount of ice this freezer could make in an hour, starting with water at °C?
2354views - Textbook Question
A certain brand of freezer is advertised to use kWh of energy per year. Assuming the freezer operates for hours each day, how much power does it require while operating?
1491views - Textbook Question
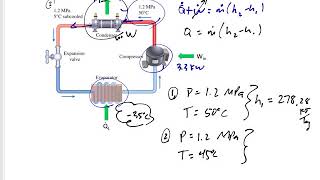
A refrigerator has a coefficient of performance of , runs on an input of W of electrical power, and keeps its inside compartment at °C. If you put a dozen -L plastic bottles of water at °C into this refrigerator, how long will it take for them to be cooled down to °C? (Ignore any heat that leaves the plastic.)
3327views - Textbook Question
The coefficient of performance is a dimensionless quantity. Its value is independent of the units used for and , as long as the same units, such as watts, are used for both quantities. However, it is common practice to express in Btu/h and in watts. When these mixed units are used, the ratio is called the energy efficiency ratio (). If a room air conditioner has , what is its ?
2034views - Multiple Choice
In the context of refrigerators, what does the coefficient of performance () represent?
59views