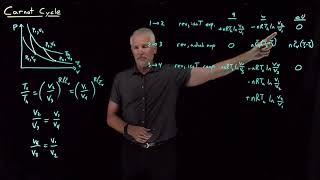23. The Second Law of Thermodynamics
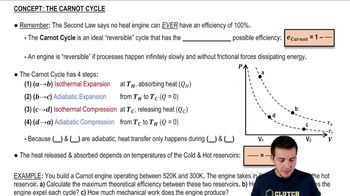
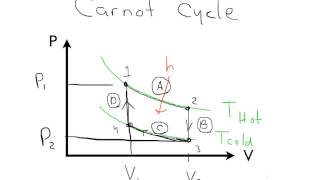
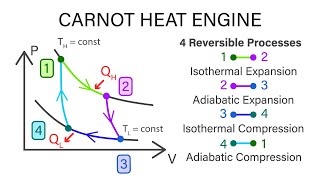
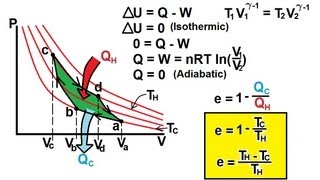
The Carnot Cycle
Learn with other creators
Practice this topic
- Multiple Choice
A theoretical heat engine in space could operate between the Sun's 5500°C surface and the –270.3°C temperature of intergalactic space. What would be its maximum theoretical efficiency?
1260views6rank1comments - Multiple Choice
A Carnot engine with an efficiency of 70% is cooled by water at 10°C. What temperature must the hot reservoir be maintained at?
2265views3rank - Multiple Choice
Your friend claims they have a design for a reversible heat engine that can operate between the freezing and boiling temperatures of water that has an efficiency of 30%. Is this possible?
1092views3rank - Multiple ChoiceWhat is the Carnot efficiency of a heat engine operating between a hot reservoir at 300°C and a cold reservoir at 10°C?1222views
- Textbook Question
A Carnot heat engine uses a hot reservoir consisting of a large amount of boiling water and a cold reservoir consisting of a large tub of ice and water. In minutes of operation, the heat rejected by the engine melts kg of ice. During this time, how much work is performed by the engine?
2201views - Textbook Question
A Carnot engine is operated between two heat reservoirs at temperatures of K and K. What is the thermal efficiency of the engine?
1163views - Textbook Question
A Carnot engine is operated between two heat reservoirs at temperatures of K and K. How much mechanical work is performed by the engine during each cycle?
1674views - Textbook Question
A Carnot engine is operated between two heat reservoirs at temperatures of K and K. If the engine receives kJ of heat energy from the reservoir at K in each cycle, how many joules per cycle does it discard to the reservoir at K?
2381views