Write balanced molecular and net ionic equations for the reaction between nitric acid and calcium hydroxide.
Ch.4 - Chemical Quantities & Aqueous Reactions

Chapter 4, Problem 87
Write balanced complete ionic and net ionic equations for each acid–base reaction. a. HBr(aq) + NaOH(aq) → b. HF(aq) + NaOH(aq) → c. HC2H3O2(aq) + RbOH(aq) →
 Verified step by step guidance
Verified step by step guidance1
Identify the type of reaction: These are acid-base reactions where an acid reacts with a base to form water and a salt.
Write the balanced molecular equation for each reaction: a. HBr(aq) + NaOH(aq) → NaBr(aq) + H₂O(l), b. HF(aq) + NaOH(aq) → NaF(aq) + H₂O(l), c. HC₂H₃O₂(aq) + RbOH(aq) → RbC₂H₃O₂(aq) + H₂O(l).
Write the complete ionic equation by dissociating all strong electrolytes into their ions: a. H⁺(aq) + Br⁻(aq) + Na⁺(aq) + OH⁻(aq) → Na⁺(aq) + Br⁻(aq) + H₂O(l), b. HF(aq) + Na⁺(aq) + OH⁻(aq) → Na⁺(aq) + F⁻(aq) + H₂O(l), c. HC₂H₃O₂(aq) + Rb⁺(aq) + OH⁻(aq) → Rb⁺(aq) + C₂H₃O₂⁻(aq) + H₂O(l).
Identify and cancel out the spectator ions that appear on both sides of the complete ionic equation: a. Na⁺ and Br⁻ are spectator ions, b. Na⁺ is a spectator ion, c. Rb⁺ is a spectator ion.
Write the net ionic equation by removing the spectator ions: a. H⁺(aq) + OH⁻(aq) → H₂O(l), b. HF(aq) + OH⁻(aq) → F⁻(aq) + H₂O(l), c. HC₂H₃O₂(aq) + OH⁻(aq) → C₂H₃O₂⁻(aq) + H₂O(l).

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
3mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Acid-Base Reactions
Acid-base reactions involve the transfer of protons (H⁺ ions) between reactants. In these reactions, acids donate protons while bases accept them. Understanding the nature of the acids and bases involved is crucial for predicting the products and writing the corresponding equations.
Recommended video:
Guided course
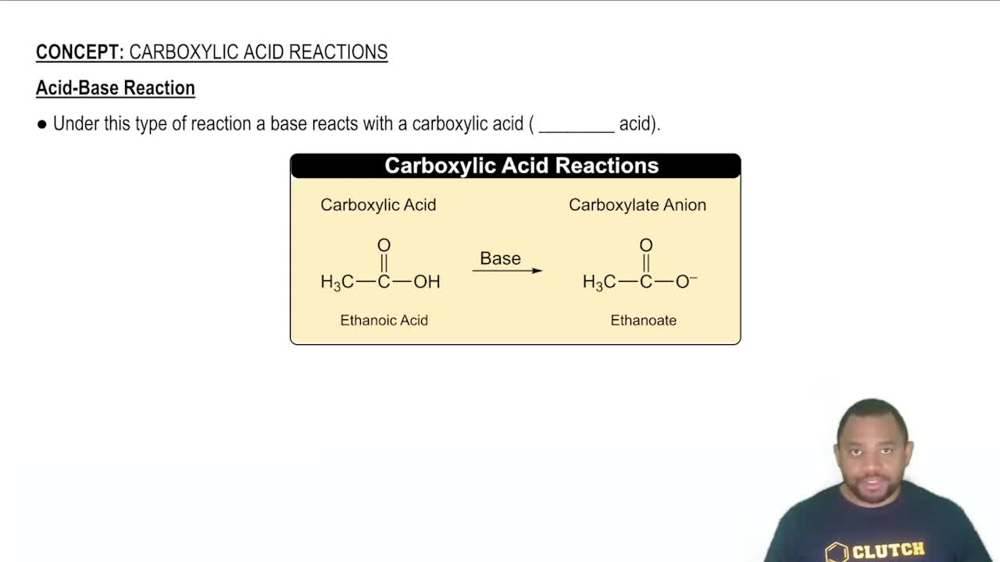
Acid-Base Reaction
Complete Ionic Equations
A complete ionic equation represents all the ions present in a solution during a reaction. It shows the reactants and products in their ionic forms, highlighting the dissociation of soluble ionic compounds. This step is essential for identifying the species that participate in the reaction.
Recommended video:
Guided course

Complete Ionic Equations
Net Ionic Equations
Net ionic equations simplify complete ionic equations by removing spectator ions—ions that do not participate in the reaction. This focuses on the actual chemical change occurring, making it easier to understand the core interaction between the reacting species. Writing net ionic equations is key for analyzing the essence of acid-base reactions.
Recommended video:
Guided course

Net Ionic Equations
Related Practice
Textbook Question
711
views
Textbook Question
Complete and balance each acid–base equation.
a. H2SO4(aq) + Ca(OH)2(aq) →
b. HClO4(aq) + KOH(aq) →
c. H2SO4(aq) + NaOH(aq) →
1370
views
Textbook Question
Complete and balance each acid–base equation. b. HC2H3O2(aq) + Ca(OH)2(aq) →
1140
views
Textbook Question
Write balanced complete ionic and net ionic equations for each acid–base reaction. a. HI(aq) + RbOH(aq) →
1345
views
Textbook Question
Write balanced complete ionic and net ionic equations for each acid–base reaction.
b. HCHO2(aq) + NaOH(aq) →
c. HC2H3O2(aq) + LiOH(aq) →
991
views
Textbook Question
A 25.00-mL sample of an unknown HClO4 solution requires titration with 22.62 mL of 0.2000 M NaOH to reach the equivalence point. What is the concentration of the unknown HClO4 solution? The neutralization reaction is HClO4(aq) + NaOH(aq) → H2O(l) + NaClO4(aq)
2379
views
1
rank
1
comments
