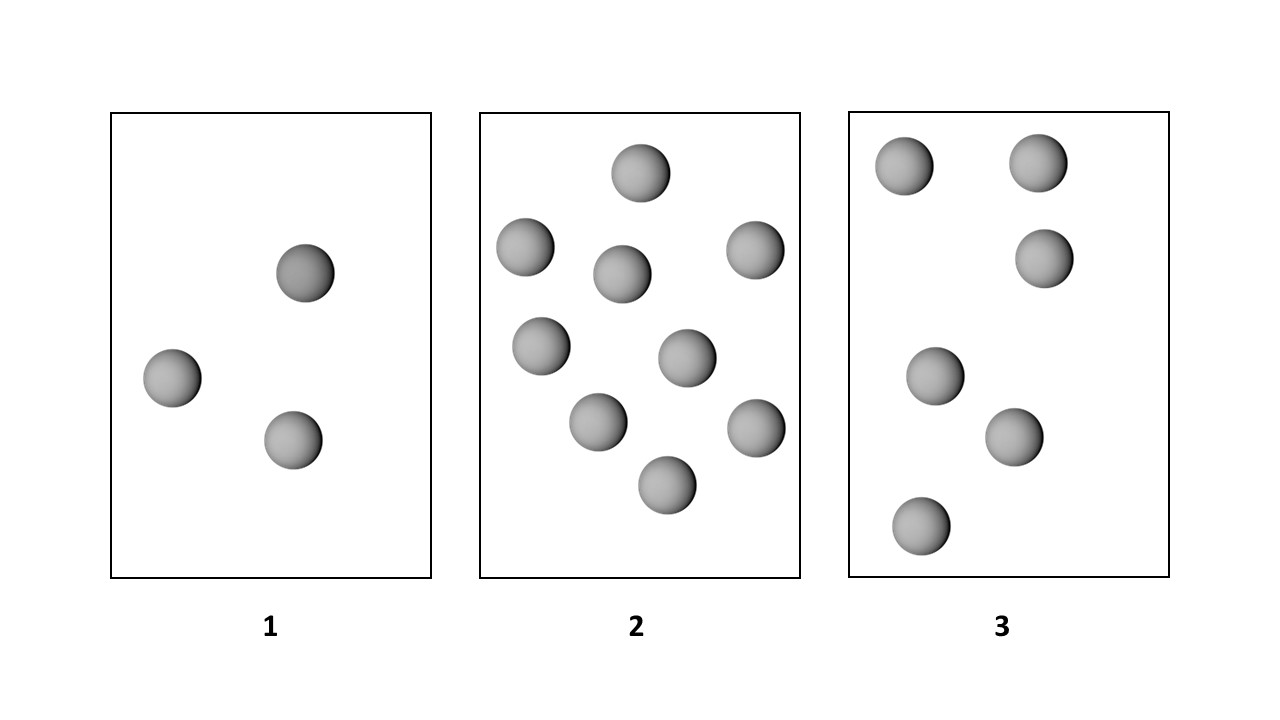
Chapter 14, Problem 41
Consider the first-order decomposition of A molecules (red spheres) in three vessels of equal volume. (1)-(3)
 Verified Solution
Verified SolutionVideo transcript
The following reaction is first order in A (red spheres) and first order in B (blue spheres): A + B → Products Rate = k[A][B]
(a) What are the relative rates of this reaction in vessels (1)–(4)? Each vessel has the same volume.
The following reaction is first order in A (red spheres) and first order in B (blue spheres): A + B → Products Rate = k[A][B]
(b) What are the relative values of the rate constant k for vessels (1)–(4)?
Consider the first-order decomposition of A molecules (red spheres) in three vessels of equal volume. (1)-(3)
(b) What are the relative half-lives of the reactions in vessels (1)–(3)?
The following pictures represent the progress of the reaction AS B in which A molecules (red spheres) are converted to B molecules (blue spheres).
(b) Draw a picture that shows the number of A and B molecules present at t = 3 min.
