(a) Place the following substances in order of increasing volatility: CH4, CBr4, CH2Cl2, CH3Cl, CHBr3, and CH2Br2. (b) How do the boiling points vary through this series? (c) Explain your answer to part (b) in terms of intermolecular forces.
Using the vapor-pressure curves in Figure 11.25, (d) estimate the external pressure at which diethyl ether will boil at 40 °C.

Verified Solution
Key Concepts
Vapor Pressure

Boiling Point

Phase Equilibrium
(a)Two pans of water are on different burners of a stove.One pan of water is boiling vigorously, while the other is boiling gently. What can be said about the temperature of the water in the two pans?
You are high up in the mountains and boil water to make some tea. However, when you drink your tea, it is not as hot as it should be. You try again and again, but the water is just not hot enough to make a hot cup of tea. Which is the best explanation for this result? (a) High in the mountains, it is probably very dry, and so the water is rapidly evaporating from your cup and cooling it. (b) High in the mountains, it is probably very windy, and so the water is rapidly evaporating from your cup and cooling it. (c) High in the mountains, the air pressure is significantly less than 1 atm, so the boiling point of water is much lower than at sea level. (d) High in the mountains, the air pressure is significantly less than 1 atm, so the boiling point of water is much higher than at sea level.
Appendix B lists the vapor pressure of water at various external pressures. (c) A city at an altitude of 5000 ft above sea level has a barometric pressure of 633 torr. To what temperature would you have to heat water to boil it in this city?
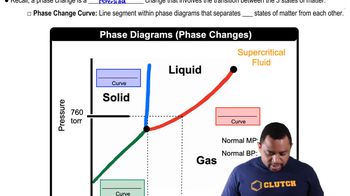
Referring to Figure 11.29, describe the phase changes (and the temperatures at which they occur) when CO2 is heated from -80 to -20°C at (a) a constant pressure of 3 atm,
