Appendix B lists the vapor pressure of water at various external pressures. (c) A city at an altitude of 5000 ft above sea level has a barometric pressure of 633 torr. To what temperature would you have to heat water to boil it in this city?
Ch.11 - Liquids and Intermolecular Forces
Chapter 11, Problem 60a
Referring to Figure 11.29, describe the phase changes (and the temperatures at which they occur) when CO2 is heated from -80 to -20°C at (a) a constant pressure of 3 atm,
 Verified step by step guidance
Verified step by step guidance1
Identify the phase diagram for CO2, which shows the phases (solid, liquid, gas) and the lines representing phase changes at different pressures and temperatures.
Locate the pressure of 3 atm on the phase diagram. This will help determine the phase changes CO2 undergoes as it is heated.
Trace a horizontal line at 3 atm from -80°C to -20°C on the phase diagram to observe the phase changes.
Note the temperatures at which the line crosses the phase boundaries. These are the temperatures at which phase changes occur.
Describe the phase changes: starting from solid CO2 (dry ice) at -80°C, identify if it sublimates directly to gas or if it passes through a liquid phase, based on the phase diagram at 3 atm.

Verified Solution
Video duration:
1mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Phase Changes
Phase changes refer to the transitions between different states of matter, such as solid, liquid, and gas. In the context of CO2, these changes include sublimation (solid to gas) and deposition (gas to solid). Understanding these transitions is crucial for analyzing how CO2 behaves under varying temperature and pressure conditions.
Recommended video:
Guided course

Entropy in Phase Changes
Phase Diagrams
A phase diagram is a graphical representation that shows the phases of a substance at different temperatures and pressures. It helps predict the state of a substance under specific conditions. For CO2, the phase diagram indicates the boundaries between solid, liquid, and gas phases, which are essential for determining the phase changes occurring at 3 atm pressure.
Recommended video:
Guided course
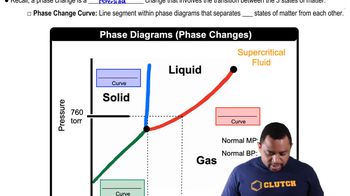
Phase Changes in Diagrams
Critical Point
The critical point is the temperature and pressure at which the distinction between liquid and gas phases disappears. For CO2, this point is significant because it defines the conditions under which CO2 can exist as a supercritical fluid. Understanding the critical point is important for predicting the behavior of CO2 when heated from -80 to -20°C at a constant pressure of 3 atm.
Recommended video:
Guided course

Boiling Point Elevation
Related Practice
Textbook Question
759
views
Open Question
What is the significance of the critical point in a phase diagram? Why does the line that separates the gas and liquid phases end at the critical point?
Textbook Question
(b) Could you measure the triple point of water by measuring the temperature in a vessel in which water vapor, liquid water, and ice are in equilibrium under 1 atm of air? Explain.
690
views
Textbook Question
The phase diagram for neon is
Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point?
1267
views
Textbook Question
Use the phase diagram of neon to answer the following questions. (b) What can you say about the strength of the intermolecular forces in neon and argon based on the critical points of Ne and Ar (see Table 11.5.)?
687
views
Textbook Question
At 25 °C gallium is a solid with a density of 5.91 g/cm3. Its melting point, 29.8 °C, is low enough that you can melt it by holding it in your hand. The density of liquid gallium just above the melting point is 6.1 g/cm3. Based on this information, what unusual feature would you expect to find in the phase diagram of gallium?
1214
views
