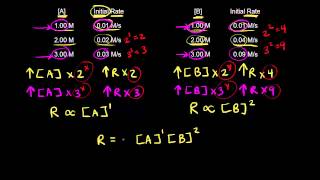15. Chemical Kinetics
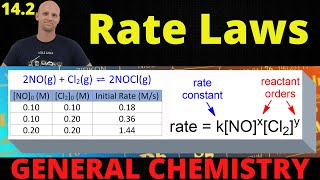
Rate Law
Problem 48
Textbook Question
Textbook QuestionConsider a reaction that occurs by the following mechanism: A + BC SAC + B AC + D SA + CD The potential energy profile for this reaction is as follows:

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
350
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos