17. Acid and Base Equilibrium
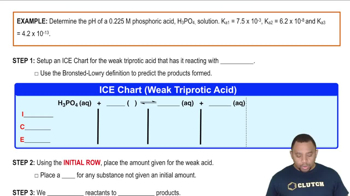
Triprotic Acids and Bases Calculations
Problem 17-148e
Textbook Question
Textbook QuestionA 40.0 mL sample of a mixture of HCl and H3PO4 was titrated with 0.100 M NaOH. The first equivalence point was reached after 88.0 mL of base, and the second equivalence point was reached after 126.4 mL of base.
(e) Sketch the pH titration curve, and label the buffer regions and equivalence points.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
82
views
Was this helpful?
Related Videos
Related Practice