17. Acid and Base Equilibrium
pH of Weak Bases
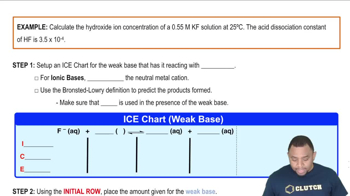
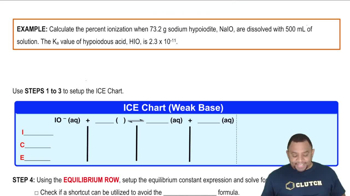
Problem 145
Textbook Question
Textbook QuestionLactic acid is a weak acid found in milk. Its calcium salt is a source of calcium for growing animals. A saturated solution of this salt, which we can represent as Ca(Lact)2, has a [Ca2 + ] = 0.26 M and a pH = 8.78. Assuming the salt is completely dissociated, calculate the Ka of lactic acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
2869
views
Was this helpful?
Related Videos
Related Practice