Consider the following reaction between oxides of nitrogen: NO2(g) + N2O(g) → 3 NO(g) (c) Calculate ΔG at 1000 K. Is the reaction spontaneous under standard conditions at this temperature?
Ch.19 - Chemical Thermodynamics

Brown14th EditionChemistry: The Central ScienceISBN: 9780134414232Not the one you use?Change textbook
Chapter 19, Problem 72
(a) Using data in Appendix C, estimate the temperature at which the free-energy change for the transformation from I2(s) to I2(g) is zero. (b) Use a reference source, such as Web Elements (www.webelements.com), to find the experimental melting and boiling points of I2. (c) Which of the values in part (b) is closer to the value you obtained in part (a)?
 Verified step by step guidance
Verified step by step guidance1
1. The free-energy change (ΔG) for a transformation is given by the equation ΔG = ΔH - TΔS, where ΔH is the enthalpy change, T is the temperature in Kelvin, and ΔS is the entropy change. For the transformation from I2(s) to I2(g), we want to find the temperature at which ΔG = 0. This occurs when ΔH = TΔS.
2. Look up the values of ΔH and ΔS for the transformation from I2(s) to I2(g) in Appendix C or a similar reference. These values are typically given in kJ/mol for ΔH and J/(mol·K) for ΔS. Note that you may need to convert the units of ΔH to J/mol to match the units of ΔS.
3. Rearrange the equation from step 1 to solve for T: T = ΔH / ΔS. Substitute the values you found in step 2 into this equation to find the temperature at which the free-energy change for the transformation is zero.
4. For part (b), use a reference source such as Web Elements to find the experimental melting and boiling points of I2. These values are typically given in degrees Celsius, so you may need to convert them to Kelvin by adding 273.15.
5. For part (c), compare the temperature you calculated in step 3 with the melting and boiling points you found in step 4. The value that is closer to the temperature you calculated is the phase transition that occurs at approximately the same temperature as the free-energy change becoming zero.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
5mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Gibbs Free Energy
Gibbs free energy (G) is a thermodynamic potential that measures the maximum reversible work obtainable from a thermodynamic system at constant temperature and pressure. The change in Gibbs free energy (ΔG) during a process indicates whether it is spontaneous; a ΔG of zero signifies equilibrium. Understanding how to calculate ΔG is essential for determining the temperature at which phase changes occur, such as the transition from solid to gas.
Recommended video:
Guided course

Gibbs Free Energy of Reactions
Phase Transition
A phase transition is the transformation of a substance from one state of matter to another, such as from solid to gas. During this process, energy is absorbed or released, affecting the temperature and pressure conditions. The melting point and boiling point are critical temperatures at which these transitions occur, and they are influenced by the substance's intermolecular forces and environmental conditions.
Recommended video:
Guided course
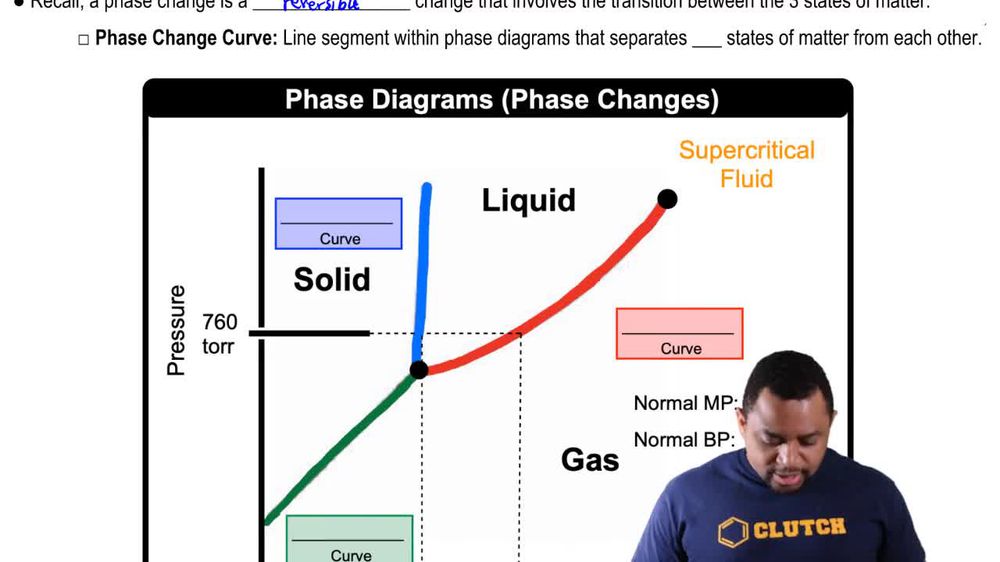
Phase Changes in Diagrams
Melting and Boiling Points
The melting point is the temperature at which a solid becomes a liquid, while the boiling point is the temperature at which a liquid becomes a gas. These points are specific to each substance and are determined by the balance of kinetic energy and intermolecular forces. Comparing the calculated temperature from part (a) with the experimental values from part (b) helps assess the accuracy of the theoretical estimation and the behavior of iodine under different conditions.
Recommended video:
Guided course
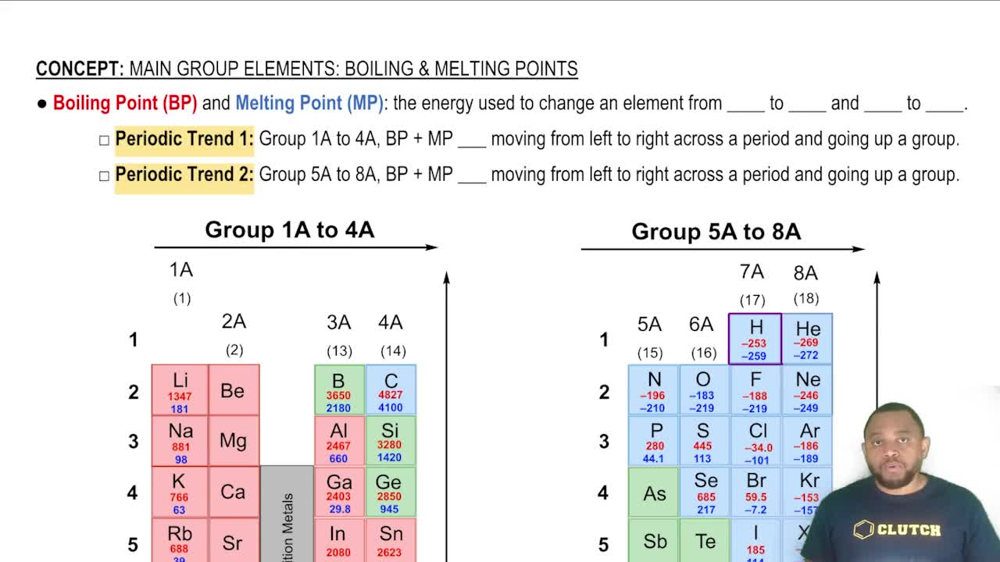
Boiling Point and Melting Point
Related Practice
Textbook Question
334
views
Textbook Question
Methanol (CH3OH) can be made by the controlled oxidation of methane: CH4(g) + 12 O2(g) → CH3OH(g) (b) Will ΔG for the reaction increase, decrease, or stay unchanged with increasing temperature?
670
views
Textbook Question
The fuel in high-efficiency natural-gas vehicles consists primarily of methane (CH4). (a) How much heat is produced in burning 1 mol of CH4(g) under standard conditions if reactants and products are brought to 298 K and H2O(l) is formed?
528
views
Textbook Question
The fuel in high-efficiency natural-gas vehicles consists primarily of methane (CH4). (b) What is the maximum amount of useful work that can be accomplished under standard conditions by this system?
558
views
