17. Acid and Base Equilibrium
Ka and Kb
Problem 114
Textbook Question
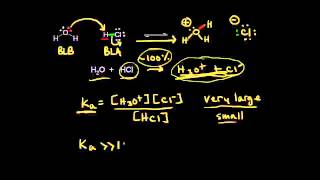
Textbook QuestionThe amino acid glycine 1H2N¬CH2¬COOH2 can participate in the following equilibria in water: H2N¬CH2¬COOH + H2OΔ H2N¬CH2¬COO- + H3O+ Ka = 4.3 * 10-3 H2N¬CH2¬COOH + H2OΔ+H3N¬CH2¬COOH + OH- Kb = 6.0 * 10-5 (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer to form a zwitterion: H2N¬CH2¬COOH Δ +H3N¬CH2¬COO-
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
872
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos