Which type (or types) of crystalline solid is characterized by each of the following? (d) network of covalent bonds.

Indicate the type of solid (molecular, metallic, ionic, or covalent-network) for each compound: (c) Ta2O5 (melting point, 1872°C)
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Types of Solids
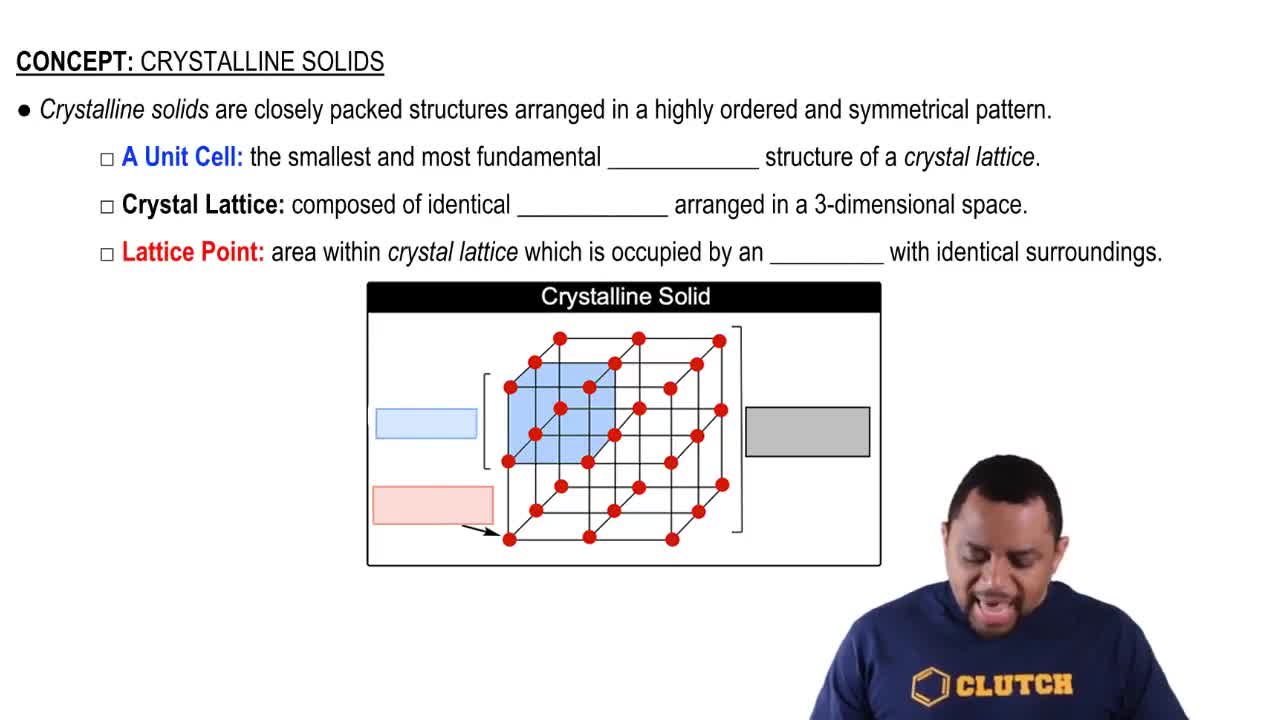
Ionic Compounds

Melting Point and Bonding
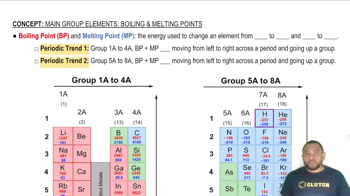
Which type (or types) of crystalline solid is characterized by each of the following? (c) high melting point and poor electrical conductivity;
Indicate the type of solid (molecular, metallic, ionic, or covalent-network) for each compound: (a) CaSO4 (molecular, metallic, ionic, or covalent-network) for each compound: (b) Pd (molecular, metallic, ionic, or covalent-network) for each compound: (d) caffeine (C8H10N4O2) (molecular, metallic, ionic, or covalent-network) for each compound: (e) toluene (C7H8) (molecular, metallic, ionic, or covalent-network) for each compound: (f) P4.
You are given a gray substance that melts at 700 °C; the solid is a conductor of electricity and is insoluble in water. Which type of solid (molecular, metallic, covalent-network, or ionic) might this substance be?
