17. Acid and Base Equilibrium
pH of Weak Bases
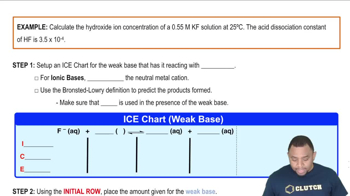
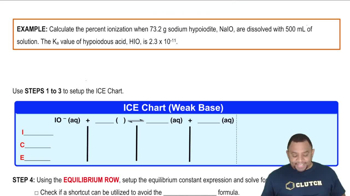
Problem 154
Textbook Question
Textbook QuestionIn aqueous solution, sodium acetate behaves as a strong electrolyte, yielding Na+ cations and CH3CO2 - anions. A particular solution of sodium acetate has a pH of 9.07 and a density of 1.0085 g/mL. What is the molality of this solution, and what is its freezing point?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
25mPlay a video:
1259
views
Was this helpful?
Related Videos
Related Practice