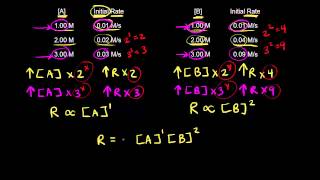15. Chemical Kinetics
Rate Law
Problem 39a
Textbook Question
Textbook QuestionA reaction in which A, B, and C react to form products is first order in A, second order in B, and zero order in C c. By what factor does the reaction rate change if [A] is doubled (and the other reactant concentrations are held constant)?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
843
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos