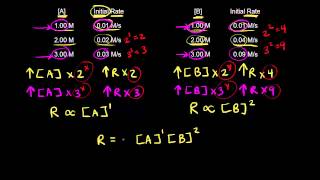15. Chemical Kinetics
Rate Law
Problem 35c
Textbook Question
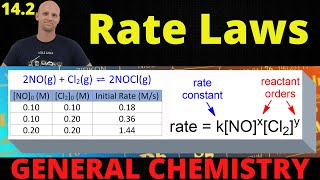
Textbook QuestionThe following data were measured for the reaction BF31g2 + NH31g2 ¡ F3BNH31g2: Experiment 3bF3 4 1M 2 3nH3 4 1M 2 Initial Rate 1M,s2 1 0.250 0.250 0.2130 2 0.250 0.125 0.1065 3 0.200 0.100 0.0682 4 0.350 0.100 0.1193 5 0.175 0.100 0.0596 (c) Calculate the rate constant with proper units?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
695
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos