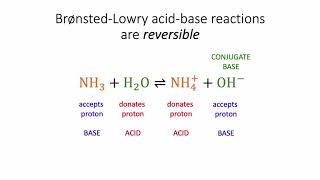17. Acid and Base Equilibrium
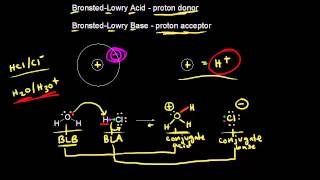
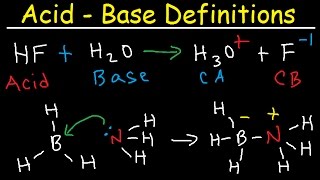
Bronsted-Lowry Acids and Bases
Problem 152
Textbook Question
Textbook QuestionIn qualitative analysis, Ca2+ and Ba2+ are seperated from Na+, K+, Mg2+ by adding aqueous (NH4)2CO3 to a solution that also contains aqueous NH3 (Figure 17.18). Assume that the concentrations after mixing are 0.080 M (NH4)2CO3 and 0.16 M NH3. (a) List all the Bronsted-Lowry acids and based present initially, and identify the principal reaction.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
516
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos