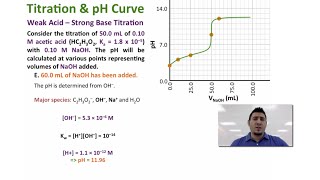
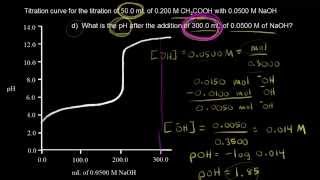
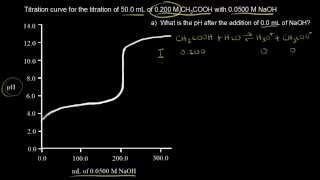
18. Aqueous Equilibrium
Titrations: Weak Acid-Strong Base
Problem 55
Textbook Question
Textbook QuestionBlood is buffered by carbonic acid and the bicarbonate ion. Normal blood plasma is 0.024 M in HCO3- and 0.0012 M H2CO3 (pKa1 for H2CO3 at body temperature is 6.1). c. Given the volume from part (b), what mass of NaOH can be neutralized before the pH rises above 7.8?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
558
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos