A 100.0 mL sample of a solution that is 0.100 M in HCl and 0.100 M in HCN is titrated with 0.100 M NaOH. Calculate the pH after the addition of the following volumes of NaOH:
(a) 0.0 mL




A 100.0 mL sample of a solution that is 0.100 M in HCl and 0.100 M in HCN is titrated with 0.100 M NaOH. Calculate the pH after the addition of the following volumes of NaOH:
(a) 0.0 mL
A 40.0 mL sample of a mixture of HCl and H3PO4 was titrated with 0.100 M NaOH. The first equivalence point was reached after 88.0 mL of base, and the second equivalence point was reached after 126.4 mL of base.
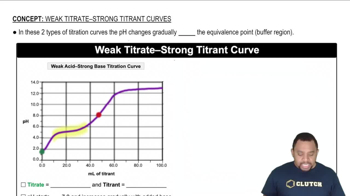
(e) Sketch the pH titration curve, and label the buffer regions and equivalence points.
The following pictures represent aqueous solutions of three acids HA1A = X, Y, or Z2; water molecules have been omitted for clarity.
(c) Which acid, if any, is a strong acid?
Which of the following pictures represents a solution of a weak diprotic acid, H2A? (Water molecules have been omitted for clarity.) Which pictures represent an impossible situation? Explain.
(a) (b) (c) (d)
The following pictures represent solutions of three salts NaA (A- = X-, Y-, or Z-); water molecules and Na+ ions have been omitted for clarity.
(a) Arrange the three A- anions in order of increasing base strength.