Here are the essential concepts you must grasp in order to answer the question correctly.
Raoult's Law
Raoult's Law states that the vapor pressure of a solvent in a solution is directly proportional to the mole fraction of the solvent in the solution. This principle is essential for calculating the vapor pressure of a solution, as it allows us to determine how the presence of a solute affects the overall vapor pressure compared to that of the pure solvent.
Recommended video:
Raoult's Law and Vapor Pressure
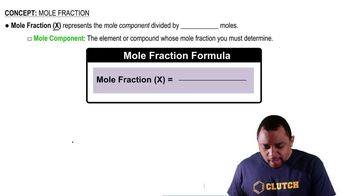
Mole Fraction
Mole fraction is a way of expressing the concentration of a component in a mixture, defined as the number of moles of that component divided by the total number of moles of all components in the mixture. In this problem, calculating the mole fractions of chloroform and acetone is crucial for applying Raoult's Law to find the vapor pressure of the solution.
Recommended video:
Ideal Solution Behavior
An ideal solution is one where the interactions between different molecules are similar to those between like molecules, leading to predictable behavior according to Raoult's Law. This assumption simplifies calculations, as it allows us to use the pure component vapor pressures and mole fractions to determine the vapor pressure of the solution without accounting for deviations due to intermolecular forces.
Recommended video: