Ethyl acetate, C4H8O2, is a fragrant substance used both as a solvent and as an aroma enhancer. Its Lewis structure is
(c) How many of the valence electrons are used to make s bonds in the molecule?
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
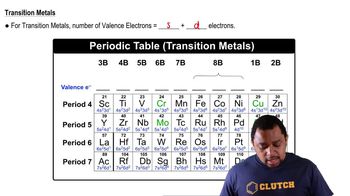
Valence Electrons
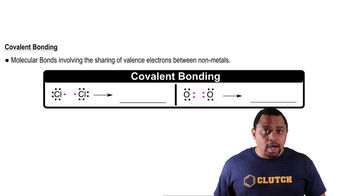
Covalent Bonds
Lewis Structure

Propylene, C3H6, is a gas that is used to form the important polymer called polypropylene. Its Lewis structure is (a) What is the total number of valence electrons in the propylene molecule?
Benzaldehyde, C7H6O, is a fragrant substance responsible for the aroma of almonds. Its Lewis structure is
(a) What is the hybridization at each of the carbon atoms of the molecule?
Ethyl acetate, C4H8O2, is a fragrant substance used both as a solvent and as an aroma enhancer. Its Lewis structure is
(e) How many valence electrons remain in nonbonding pairs in the molecule?
Consider the Lewis structure for acetic acid, which is known as vinegar:
(a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each of them?
Consider the Lewis structure for glycine, the simplest amino acid:
(b) What are the hybridizations of the orbitals on the two oxygens and the nitrogen atom, and what are the approximate bond angles at the nitrogen?
