Here are the essential concepts you must grasp in order to answer the question correctly.
pH Scale
The pH scale measures the acidity or basicity of a solution, ranging from 0 to 14. A pH of 7 is neutral, below 7 indicates acidity, and above 7 indicates basicity. The scale is logarithmic, meaning each whole number change represents a tenfold change in hydrogen ion concentration.
Recommended video:
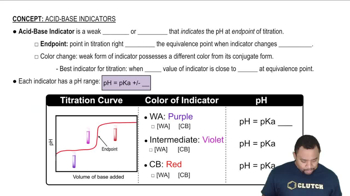
Indicators and Their pH Ranges
Indicators are substances that change color in response to pH changes. Methyl orange transitions from red to yellow between pH 3.1 and 4.4, while bromthymol blue changes from yellow to blue between pH 6.0 and 7.6. The color change indicates the pH range of the solution based on the indicator used.
Recommended video:
Acid-Base Properties of Solutions
The acid-base properties of a solution determine its behavior with indicators. A solution that turns both methyl orange and bromthymol blue yellow suggests it is likely acidic but not strongly so, indicating a pH below 6.0. This information helps narrow down the possible pH range for the unknown solution.
Recommended video:
Arrhenius Acids and Bases
 Verified step by step guidance
Verified step by step guidance