Open Question
Carbon dioxide in the atmosphere dissolves in raindrops to produce carbonic acid (H2CO3), causing the pH of clean, unpolluted rain to range from about 5.2 to 5.6. What are the ranges of [H+] and [OH-] in the raindrops?
 Verified step by step guidance
Verified step by step guidance
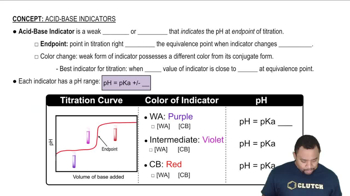


Addition of the indicator methyl orange to an unknown solution leads to a yellow color. The addition of bromthymol blue to the same solution also leads to a yellow color. (b) What is the range (in whole numbers) of possible pH values for the solution?
Addition of phenolphthalein to an unknown colorless solution does not cause a color change. The addition of bromthymol blue to the same solution leads to a yellow color. (b) Which of the following can you establish about the solution: (i) A minimum pH, (ii) A maximum pH, or (iii) A specific range of pH values?
Calculate the pH of each of the following strong acid solutions: (b) 1.52 g of HNO3 in 575 mL of solution