(c) Which step is the fastest?
Consider the following energy profile.
(c) Which step is rate limiting?
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Energy Profile Diagram

Activation Energy
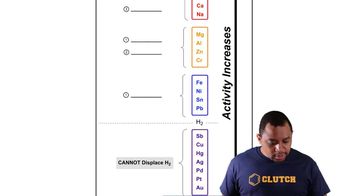
Rate-Limiting Step

Consider the following energy profile.
(a) How many elementary reactions are in the reaction mechanism?
Consider the following energy profile.
(b) How many intermediates are formed in the reaction?
The decomposition of hydrogen peroxide is catalyzed by iodide ion. The catalyzed reaction is thought to proceed by a two-step mechanism:
H2O2(aq) + I-(aq) → H2O(l) + IO-(aq) (slow)
IO-(aq) + H2O2(aq) → H2O(l) + O2(g) + I-(aq) (fast)
(a) Write the chemical equation for the overall process.
The decomposition of hydrogen peroxide is catalyzed by iodide ion. The catalyzed reaction is thought to proceed by a two-step mechanism:
H2O2(aq) + I-(aq) → H2O(l) + IO-(aq) (slow)
IO-(aq) + H2O2(aq) → H2O(l) + O2(g) + I-(aq) (fast)
(b) Identify the intermediate, if any, in the mechanism.
