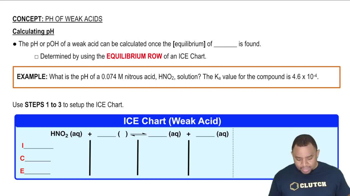
17. Acid and Base Equilibrium
pH of Weak Acids
Problem 54
Textbook Question
Textbook QuestionA 0.100 M solution of bromoacetic acid 1BrCH2COOH2 is 13.2% ionized. Calculate 3H+4, 3BrCH2COO-4, 3BrCH2COOH4 and Ka for bromoacetic acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
816
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos