15. Chemical Kinetics
Energy Diagrams
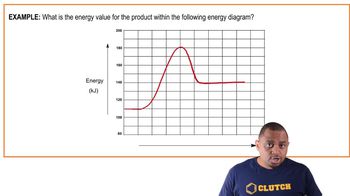
Problem 96
Textbook Question
Textbook QuestionConsider the reaction in which HCl adds across the double bond of ethene: HCl + H2C'CH2¡H3C¬CH2Cl The following mechanism, with the accompanying energy diagram, has been suggested for this reaction: Step 1 HCl + H2C'CH2¡H3C'CH2+ + Cl - Step 2 H3C'CH2+ + Cl - ¡H3C¬CH2Cl

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
858
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos