11. Bonding & Molecular Structure
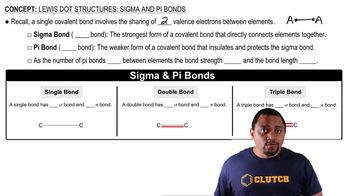
Lewis Dot Structures: Sigma & Pi Bonds
Problem 103
Textbook Question
Textbook QuestionThe structure of borazine, B3N3H6, is a six-membered ring of alternating B and N atoms. There is one H atom bonded to each B and to each N atom. The molecule is planar. (e) What are the hybridizations at the B and N atoms in the Lewis structures from parts (a) and (b)? Would you expect the molecule to be planar for both Lewis structures? Would you expect the molecule to be planar for both Lewis structures?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1720
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos