Textbook Question
Write equations for the half-reactions that occur in the electrolysis of molten potassium bromide.
1527
views
1
rank

 Verified step by step guidance
Verified step by step guidance

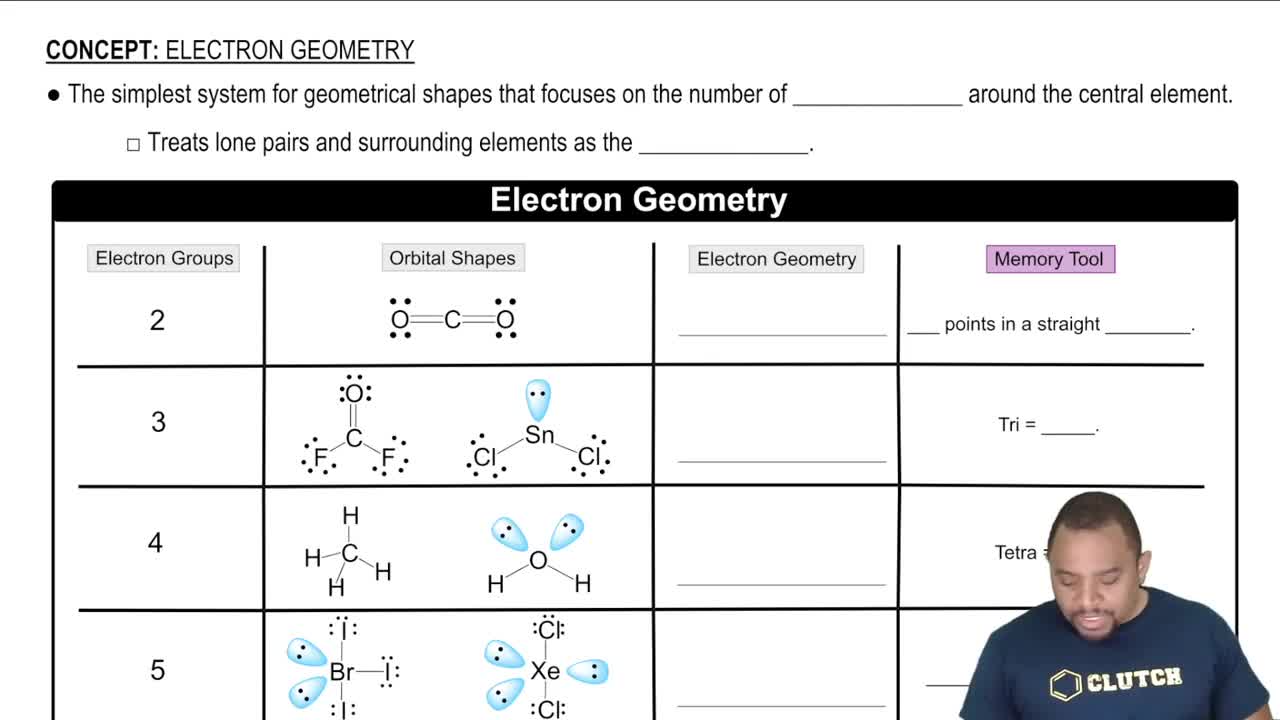
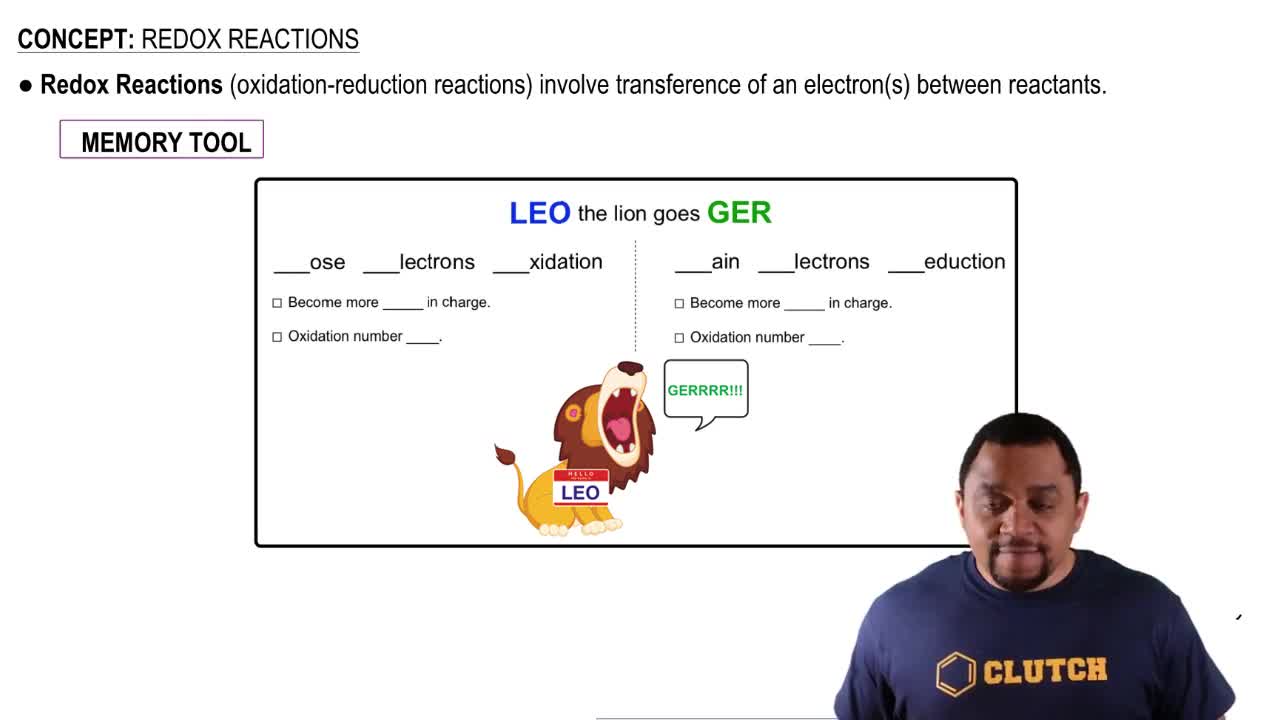
Write equations for the half-reactions that occur in the electrolysis of molten potassium bromide.
Determine whether or not each metal, if coated onto iron, would prevent the corrosion of iron. b. Cr
What products are obtained in the electrolysis of molten NaI?
Draw an electrolytic cell in which Mn2+ is reduced to Mn and Sn is oxidized to Sn2+. Label the anode and cathode, indicate the direction of electron flow, and write an equation for the half-reaction occurring at each electrode. What minimum voltage is necessary to drive the reaction?
Determine whether or not each metal, if coated onto iron, would prevent the corrosion of iron. a. Mg
Determine whether or not each metal, if coated onto iron, would prevent the corrosion of iron. c. Cu