15. Chemical Kinetics
Reaction Mechanism
Problem 107
Textbook Question
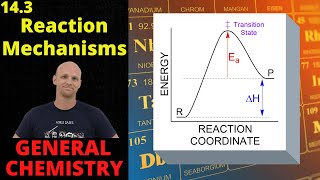
Textbook QuestionThe following mechanism has been proposed for the reaction of NO with H2 to form N2O and H2O: NO1g2 + NO1g2¡N2O21g2 N2O21g2 + H21g2¡N2O1g2 + H2O1g2 (d) The observed rate law is rate = k3NO423H24. If the proposed mechanism is correct, what can we conclude about the relative speeds of the first and second reactions?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1598
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos