9. Quantum Mechanics
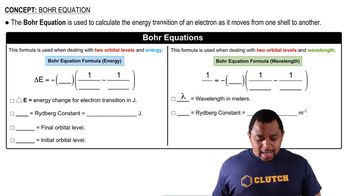
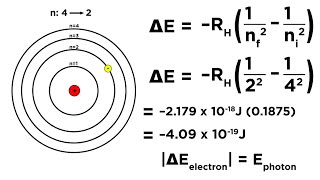
Bohr Equation
Problem 124
Textbook Question
Textbook QuestionOrbital energies in single-electron atoms or ions, such as He+, can be described with an equation similar to the Balmer–Rydberg equation:

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
677
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos