In January 2006, the New Horizons space probe was launched from Earth with the mission to perform a flyby study of Pluto. The arrival at the dwarf planet was estimated to happen after nine years, in 2015. The distance between Earth and Pluto varies depending on the location of the planets in their orbits, but at their closest, the distance is 4.2 billion kilometers (2.6 billion miles). Calculate the minimum amount of time it takes for a transmitted signal from Pluto to reach the Earth.
In an experiment to study the photoelectric effect, a scientist measures the kinetic energy of ejected electrons as a function of the frequency of radiation hitting a metal surface. She obtains the following plot. The point labeled 'n0' corresponds to light with a wavelength of 542 nm. (a) What is the value of n0 in s - 1?

 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Photoelectric Effect
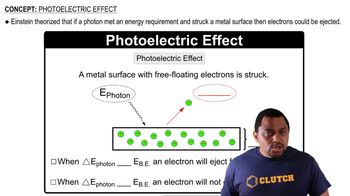
Frequency and Wavelength Relationship

Kinetic Energy of Ejected Electrons

The watt is the derived SI unit of power, the measure of energy per unit time: 1 W = 1 J>s. A semiconductor laser in a DVD player has an output wavelength of 650 nm and a power level of 5.0 mW. How many photons strike the DVD surface during the playing of a DVD 90 minutes in length?
Consider a transition in which the electron of a hydrogen atom is excited from n = 1 to n = . (a) What is the end result of this transition?
Consider a transition in which the electron of a hydrogen atom is excited from n = 1 to n = . (b) What is the wavelength of light that must be absorbed to accomplish this process?
Consider a transition in which the electron of a hydrogen atom is excited from n = 1 to n = . (d) How are the results of parts (b) and (c) related to the plot shown in Exercise 6.88?
