Textbook Question
An electron traveling at 3.7×105 m/s has an uncertainty in its velocity of 1.88×105 m/s. What is the uncertainty in its position?
2303
views
1
rank
1
comments
 Verified step by step guidance
Verified step by step guidance


An electron traveling at 3.7×105 m/s has an uncertainty in its velocity of 1.88×105 m/s. What is the uncertainty in its position?
Which electron is, on average, farther from the nucleus: an electron in a 3p orbital or an electron in a 4p orbital?
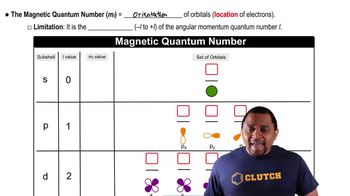
What are the possible values of l for each given value of n? a. 1 b. 2 c. 3 d. 4
What are the possible values of ml for each value of l? b. 1
What are the possible values of ml for each value of l? c. 2 d. 3
Which set of quantum numbers cannot occur together to specify an orbital? a. n = 2, l = 1, ml = -1 b. n = 3, l = 2, ml = 0 c. n = 3, l = 3, ml = 2 d. n = 4, l = 3, ml = 0