Exactly 1.5 g of a fuel burns under conditions of constant pressure and then again under conditions of constant volume. In measurement A the reaction produces 25.9 kJ of heat, and in measurement B the reaction produces 23.3 kJ of heat. Which measurement (A or B) corresponds to conditions of constant pressure? Explain.
Instant cold packs used to ice athletic injuries on the field contain ammonium nitrate and water separated by a thin plastic divider. When the divider is broken, the ammonium nitrate dissolves according to the endothermic reaction: NH4NO3(s) → NH4+(aq) + NO3– (aq) In order to measure the enthalpy change for this reaction, 1.25 g of NH4NO3 is dissolved in enough water to make 25.0 mL of solution. The initial temperature is 25.8 °C and the final temperature (after the solid dissolves) is 21.9 °C. Calculate the change in enthalpy for the reaction in kJ. (Use 1.0 g/mL as the density of the solution and 4.18 J/g•°C as the specific heat capacity.)

Verified Solution
Key Concepts
Endothermic Reactions
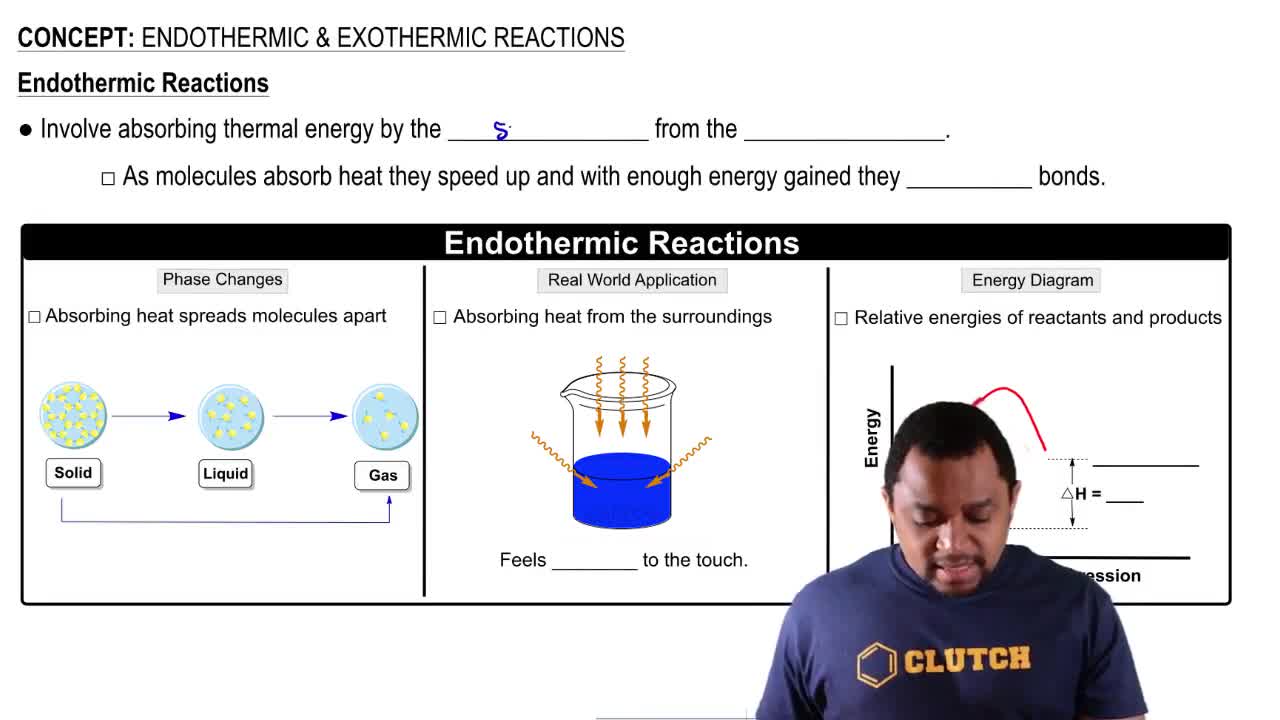
Enthalpy Change (ΔH)

Specific Heat Capacity

When 1.03 g of biphenyl (C12H10) undergoes combustion in a bomb calorimeter, the temperature rises from 24.2 °C to 2931.4 °C. Find ΔErxn for the combustion of biphenyl in kJ>mol biphenyl. The heat capacity of the bomb calorimeter, determined in a separate experiment, is 5.86 kJ/°C.
Zinc metal reacts with hydrochloric acid according to the balanced equation: Zn(s) + 2 HCl(aq) → ZnCl2(aq) + H2(g) When 0.103 g of Zn(s) is combined with enough HCl to make 50.0 mL of solution in a coffee-cup calorimeter, all of the zinc reacts, raising the temperature of the solution from 22.5 °C to 23.7 °C. Find ΔHrxn for this reaction as written. (Use 1.0 g/mL for the density of the solution and 4.18 J/g•°C as the specific heat capacity.)
For each generic reaction, determine the value of ΔH2 in terms of ΔH1.
a. A + B → 2 C ΔH1
2 C→ A + B ΔH2 = ?
For each generic reaction, determine the value of ΔH2 in terms of ΔH1.
b. A + 1/2 B → C ΔH1
2 A + B → 2 C ΔH2 = ?
For each generic reaction, determine the value of ΔH2 in terms of ΔH1.
c. A → B + 2 C ΔH1
1/2 B + C → 1/2 A ΔH2 = ?
