Which statements are consistent with Rutherford's nuclear theory as it was originally stated? Why? a. The volume of an atom is mostly empty space. b. The nucleus of an atom is small compared to the size of the atom. c. Neutral lithium atoms contain more neutrons than protons. d. Neutral lithium atoms contain more protons than electrons.
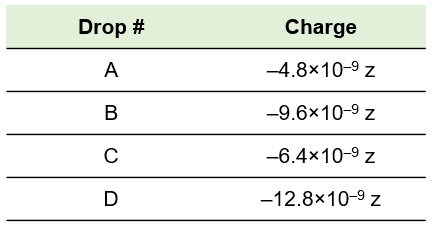
Imagine a unit of charge called the zorg. A chemist performs the oil drop experiment and measures the charge of each drop in zorgs. Based on the results shown here, what is the charge of the electron in zorgs (z)? How many electrons are in each drop?
Drop # Charge
A –4.8×10–9 z
B –9.6×10–9 z
C –6.4×10–9 z
D –12.8×10–9 z
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Charge of the Electron

Oil Drop Experiment

Quantization of Charge

Which statements are inconsistent with Rutherford's nuclear theory as it was originally stated? Why? a. Since electrons are smaller than protons, and since a hydrogen atom contains only one proton and one electron, it must follow that the volume of a hydrogen atom is mostly due to the proton. b. A nitrogen atom has 7 protons in its nucleus and 7 electrons outside of its nucleus. c. A phosphorus atom has 15 protons in its nucleus and 150 electrons outside of its nucleus. d. The majority of the mass of a fluorine atom is due to its 9 electrons.
A chemist in an imaginary universe, where electrons have a different charge than they do in our universe, performs the Millikan oil drop experiment to measure the electron's charge. The charges of several drops are recorded here. What is the charge of the electron in this imaginary universe?
Drop # Charge
A –6.9×10–19 C
B –9.2×10–19 C
C –11.5×10–19 C
D –4.6×10–19 C
On a dry day, your body can accumulate static charge from walking across a carpet or from brushing your hair. If your body develops a charge of -22 µC (microcoulombs), how many excess electrons has it acquired?
On a dry day, your body can accumulate static charge from walking across a carpet or from brushing your hair. If your body develops a charge of -22 mC (microcoulombs), what is their collective mass?
How many electrons are necessary to produce a charge of -11.0 C? What is the mass of this many electrons?
