Calculate the vapor pressure of a solution containing 24.5 g of glycerin (C3H8O3) in 135 mL of water at 30.0 °C. The vapor pressure of pure water at this temperature is 31.8 torr. Assume that glycerin is not volatile and dissolves molecularly (i.e., it is not ionic), and use a density of 1.00 g/mL for the water.
Ch.13 - Solutions
Chapter 13, Problem 74
A solution contains a mixture of pentane and hexane at room temperature. The solution has a vapor pressure of 258 torr. Pure pentane and hexane have vapor pressures of 425 torr and 151 torr, respectively, at room temperature. What is the mole fraction composition of the mixture? (Assume ideal behavior.)

Verified Solution
Video duration:
6mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Raoult's Law
Raoult's Law states that the vapor pressure of a solvent in a solution is directly proportional to the mole fraction of the solvent in the solution. For a mixture of volatile components, the total vapor pressure is the sum of the partial pressures of each component, which can be calculated using their respective mole fractions and pure component vapor pressures.
Recommended video:
Guided course

Raoult's Law and Vapor Pressure
Mole Fraction
Mole fraction is a way of expressing the concentration of a component in a mixture. It is defined as the ratio of the number of moles of a specific component to the total number of moles of all components in the mixture. This concept is crucial for applying Raoult's Law to determine the contributions of each component to the overall vapor pressure.
Recommended video:
Guided course
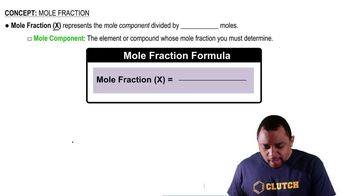
Mole Fraction Formula
Ideal Gas Behavior
Ideal gas behavior refers to the assumption that gases behave according to the ideal gas law, where interactions between gas molecules are negligible, and the volume of the gas molecules themselves is also negligible. In the context of solutions, assuming ideal behavior allows for the simplification of calculations involving vapor pressures and mole fractions, making it easier to predict the properties of the mixture.
Recommended video:
Guided course

Ideal Gas Law Formula
Related Practice
Textbook Question
3149
views
Open Question
A solution contains naphthalene (C10H8) dissolved in hexane (C6H14) at a concentration of 12.35% naphthalene by mass. Calculate the vapor pressure of hexane above the solution at 25 °C. The vapor pressure of pure hexane at 25 °C is 151 torr.
Textbook Question
A solution contains 50.0 g of heptane (C7H16) and 50.0 g of octane (C8H18) at 25 °C. The vapor pressures of pure heptane and pure octane at 25 °C are 45.8 torr and 10.9 torr, respectively. Assuming ideal behavior, answer the following: d. Why is the composition of the vapor different from the composition of the solution?
1207
views
Open Question
A solution contains 4.08 g of chloroform (CHCl3) and 9.29 g of acetone (CH3COCH3). The vapor pressures at 35 °C of pure chloroform and pure acetone are 295 torr and 332 torr, respectively. Assuming ideal behavior, calculate the vapor pressures of each of the components and the total vapor pressure above the solution. The experimentally measured total vapor pressure of the solution at 35 °C is 312 torr. Is the solution ideal? If not, what can you say about the relative strength of chloroform–acetone interactions compared to the acetone–acetone and chloroform–chloroform interactions?
Open Question
A solution of methanol and water has a mole fraction of water of 0.312 and a total vapor pressure of 211 torr at 39.9 °C. The vapor pressures of pure methanol and pure water at this temperature are 256 torr and 55.3 torr, respectively. Is the solution ideal? If not, what can be inferred about the relative strengths of the solute–solvent interactions compared to the solute–solute and solvent–solvent interactions?
Textbook Question
A glucose solution contains 55.8 g of glucose (C6H12O6) in 455 g of water. Determine the freezing point and boiling point of the solution.
3181
views
