Textbook Question
Draw a molecular orbital energy diagram for ClF. (Assume that the sp orbitals are lower in energy than the p orbitals.) What is the bond order in ClF?
1641
views
 Tro 4th Edition
Tro 4th Edition Ch.10 - Chemical Bonding II: Molecular Shapes & Valence Bond Theory
Ch.10 - Chemical Bonding II: Molecular Shapes & Valence Bond Theory Problem 94
Problem 94 Verified step by step guidance
Verified step by step guidance


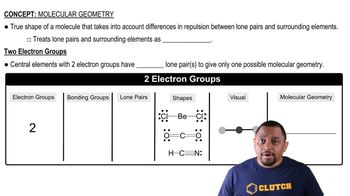
Draw a molecular orbital energy diagram for ClF. (Assume that the sp orbitals are lower in energy than the p orbitals.) What is the bond order in ClF?
Draw Lewis structures and MO diagrams for CN+ , CN, and CN- . According to the Lewis model, which species is most stable?
Draw the structure of a molecule with the formula C4H6Cl2 that has a dipole moment of 0.
Draw the structures of two compounds that have the composition CH3NO2 and have all three H atoms bonded to the C. Predict which compound has the larger ONO bond angle.
How many types of hybrid orbitals do we use to describe each molecule? a. N2O5 c. BrCN (no formal charges)