Here are the essential concepts you must grasp in order to answer the question correctly.
Hybridization
Hybridization is the concept of mixing atomic orbitals to form new hybrid orbitals that can accommodate bonding in molecules. In borazine, the boron (B) and nitrogen (N) atoms undergo sp2 hybridization, which involves the mixing of one s orbital and two p orbitals. This results in three equivalent sp2 hybrid orbitals that allow for the formation of sigma bonds with adjacent atoms.
Recommended video:
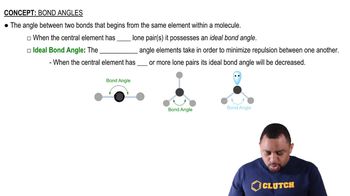
Bond Angles
Bond angles are the angles formed between adjacent bonds in a molecule, which are influenced by the hybridization of the atoms involved. In borazine, the B-N-B bond angle is approximately 120 degrees, and the N-B-N bond angle is also around 120 degrees. These angles reflect the trigonal planar arrangement of the sp2 hybridized orbitals around each atom.
Recommended video:
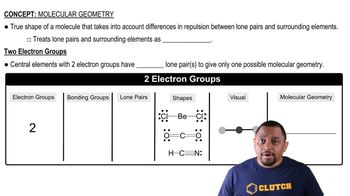
Molecular Geometry
Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule. In borazine, the overall shape is trigonal planar due to the sp2 hybridization of the B and N atoms, which allows for a flat structure. This geometry is characterized by the equal bond angles and the arrangement of the atoms in a single plane, contributing to the molecule's stability and reactivity.
Recommended video:
Molecular Geometry with Two Electron Groups