Here are the essential concepts you must grasp in order to answer the question correctly.
Isotopes
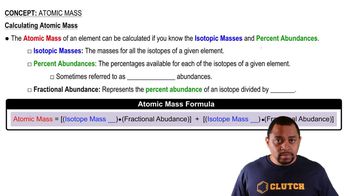
Isotopes are variants of a chemical element that have the same number of protons but different numbers of neutrons, resulting in different atomic masses. For example, copper has two isotopes, copper-63 and copper-65, which differ in their mass due to the varying number of neutrons. Understanding isotopes is crucial for calculating the average atomic weight of an element based on its isotopic composition.
Recommended video:
Atomic Weight Calculation
The atomic weight of an element is calculated as the weighted average of the masses of its isotopes, taking into account their relative abundances. This is done by multiplying the mass of each isotope by its percentage abundance (expressed as a fraction) and summing these values. For copper, the calculation involves using the isotopic masses and their respective percentages to find the overall atomic weight.
Recommended video:
Periodic Table
The periodic table is a tabular arrangement of chemical elements, organized by increasing atomic number and grouped by similar properties. Each element's atomic weight is listed, which reflects the average mass of its naturally occurring isotopes. Checking the calculated atomic weight against the periodic table helps verify the accuracy of the calculation and provides a reference for understanding the element's characteristics.
Recommended video:
Periodic Table Classifications