Chlorine gas was first prepared in 1774 by the oxidation of NaCl with MnO2: 2 NaCl(s) + 2 H2SO4(l) + MnO2(s) → Na2SO4(s) + MnSO4(s) + 2 H2O(g) + Cl2(g) Assume that the gas produced is saturated with water vapor at a partial pressure of 28.7 mm Hg and that it has a volume of 0.597 L at 27 °C and 755 mm Hg pressure. (b) How many grams of NaCl were used in the experiment, assuming complete reaction?
Natural gas is a mixture of hydrocarbons, primarily methane (CH4) and ethane (C2H6). A typical mixture might have Xmethane = 0.915 and Xethane = 0.085. Let's assume that we have a 15.50 g sample of natural gas in a volume of 15.00 L at a temperature of 20.00 °C. (c) What is the partial pressure of each component in the sample in atmospheres?

Verified Solution
Key Concepts
Dalton's Law of Partial Pressures

Ideal Gas Law

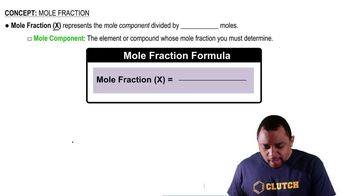
Mole Fraction
Natural gas is a mixture of hydrocarbons, primarily methane 1CH42 and ethane 1C2H62. A typical mixture might have Xmethane = 0.915 and Xethane = 0.085. Let's assume that we have a 15.50 g sample of natural gas in a volume of 15.00 L at a temperature of 20.00 °C. (a) How many total moles of gas are in the sample?
Natural gas is a mixture of hydrocarbons, primarily methane (CH4) and ethane (C2H6). A typical mixture might have Xmethane = 0.915 and Xethane = 0.085. Let's assume that we have a 15.50 g sample of natural gas in a volume of 15.00 L at a temperature of 20.00 °C. (b) What is the pressure of the sample in atmospheres?
Gaseous compound Q contains only xenon and oxygen. When 0.100 g of Q is placed in a 50.0-mL steel vessel at 0 °C the pressure is 0.229 atm. (a) What is the molar mass of Q, and what is a likely formula?
Gaseous compound Q contains only xenon and oxygen. When 0.100 g of Q is placed in a 50.0-mL steel vessel at 0 °C the pressure is 0.229 atm. (b) When the vessel and its contents are warmed to 100 °C, Q decomposes into its constituent elements. What is the total pressure, and what are the partial pressures of xenon and oxygen in the container?
