Natural gas is a mixture of hydrocarbons, primarily methane 1CH42 and ethane 1C2H62. A typical mixture might have Xmethane = 0.915 and Xethane = 0.085. Let's assume that we have a 15.50 g sample of natural gas in a volume of 15.00 L at a temperature of 20.00 °C. (a) How many total moles of gas are in the sample?

Verified Solution
Key Concepts
Molar Mass

Ideal Gas Law

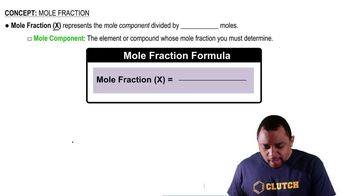
Mole Fraction
Chlorine gas was first prepared in 1774 by the oxidation of NaCl with MnO2: 2 NaCl(s) + 2 H2SO4(l) + MnO2(s) → Na2SO4(s) + MnSO4(s) + 2 H2O(g) + Cl2(g) Assume that the gas produced is saturated with water vapor at a partial pressure of 28.7 mm Hg and that it has a volume of 0.597 L at 27 °C and 755 mm Hg pressure. (a) What is the mole fraction of Cl2 in the gas?
Chlorine gas was first prepared in 1774 by the oxidation of NaCl with MnO2: 2 NaCl(s) + 2 H2SO4(l) + MnO2(s) → Na2SO4(s) + MnSO4(s) + 2 H2O(g) + Cl2(g) Assume that the gas produced is saturated with water vapor at a partial pressure of 28.7 mm Hg and that it has a volume of 0.597 L at 27 °C and 755 mm Hg pressure. (b) How many grams of NaCl were used in the experiment, assuming complete reaction?
Natural gas is a mixture of hydrocarbons, primarily methane (CH4) and ethane (C2H6). A typical mixture might have Xmethane = 0.915 and Xethane = 0.085. Let's assume that we have a 15.50 g sample of natural gas in a volume of 15.00 L at a temperature of 20.00 °C. (b) What is the pressure of the sample in atmospheres?
Natural gas is a mixture of hydrocarbons, primarily methane (CH4) and ethane (C2H6). A typical mixture might have Xmethane = 0.915 and Xethane = 0.085. Let's assume that we have a 15.50 g sample of natural gas in a volume of 15.00 L at a temperature of 20.00 °C. (c) What is the partial pressure of each component in the sample in atmospheres?
Gaseous compound Q contains only xenon and oxygen. When 0.100 g of Q is placed in a 50.0-mL steel vessel at 0 °C the pressure is 0.229 atm. (a) What is the molar mass of Q, and what is a likely formula?
